 
There are intermediate cases but these are normally treated specially. The former applies to distinguishable molecules (they can be distinguished by their position in the lattice) and in the latter the N! allows for the molecules being indistinguishable. The partition function Q is related to the molecular partition function q byįor a gas. At this level it does not need to be derived or justified in any way. This equation should be the starting point of most derivations. Where A is the Helmholtz free energy and Q is the partition function for the whole system. 
The most important way of expressing the basic assumption (often referred to as the canonical ensemble) is It is sometimes referred to as the microcanonical ensemble. residual entropies and number of ways of arranging atoms on adsorption sites (see Third Year Statistical Mechanics), but it is not generally useful (or used) for deriving more complicated relations. 
This is a useful expression where it is necessary to relate entropy to disorder, e.g. Where Ω is the number of ways of arranging the system (number of configurations, etc.). The basic assumption of statistical mechanics can be written in a number of ways. The Thomas Group - PTCL, Oxford Statistical Mechanics - 2nd YearĪs well as doing the problems below, you should revise the work on heat capacities that you were given in the first year and some of the problems set in Vibrations and Rotations. Classical discrete system įor a canonical ensemble that is classical and discrete, the canonical partition function is defined asĠ ≡ δ L = δ ( − ∑ i k B ρ i ln ρ i ) + δ ( λ 1 − ∑ i λ 1 ρ i ) + δ ( λ 2 U − ∑ i λ 2 ρ i E i ) = ∑ i = ∑ i = ∑ i δ ( ρ i ). The appropriate mathematical expression for the canonical partition function depends on the degrees of freedom of the system, whether the context is classical mechanics or quantum mechanics, and whether the spectrum of states is discrete or continuous. A collection of this kind of system comprises an ensemble called a canonical ensemble. Initially, let us assume that a thermodynamically large system is in thermal contact with the environment, with a temperature T, and both the volume of the system and the number of constituent particles are fixed. The partition function has many physical meanings, as discussed in Meaning and significance.Ĭanonical partition function Definition Other types of partition functions can be defined for different circumstances see partition function (mathematics) for generalizations. The grand canonical partition function applies to a grand canonical ensemble, in which the system can exchange both heat and particles with the environment, at fixed temperature, volume, and chemical potential. The canonical partition function applies to a canonical ensemble, in which the system is allowed to exchange heat with the environment at fixed temperature, volume, and number of particles. 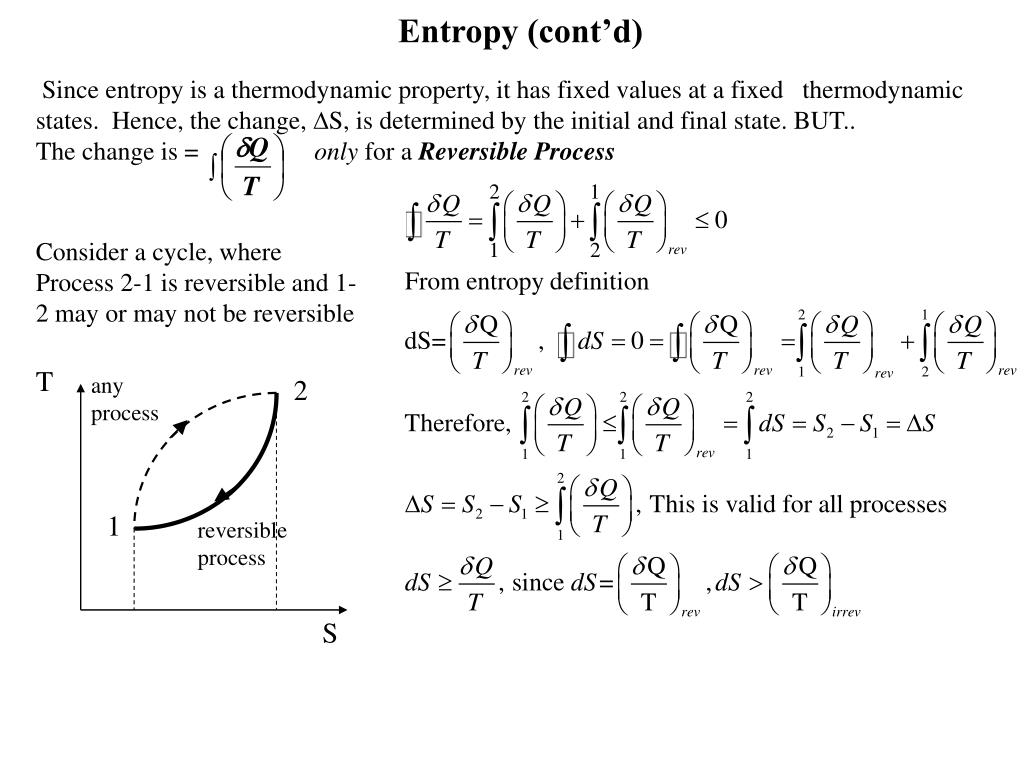
The most common statistical ensembles have named partition functions. The partition function is dimensionless.Įach partition function is constructed to represent a particular statistical ensemble (which, in turn, corresponds to a particular free energy). Most of the aggregate thermodynamic variables of the system, such as the total energy, free energy, entropy, and pressure, can be expressed in terms of the partition function or its derivatives. Partition functions are functions of the thermodynamic state variables, such as the temperature and volume. In physics, a partition function describes the statistical properties of a system in thermodynamic equilibrium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
